In this article, I will give you an insight into the pharmaceutical industry and answer the following questions:
- Why is the pharma industry such a booming market?
- What does it take to bring a medicinal product to the market?
- Why medicine costs so much?
- What is patent law, and what does it have to do with the pharmaceutical industry?
- What two main types of pharmaceutical manufacturers are there?
So let’s get right to it!
The Pharmaceutical Market
The pharmaceutical industry is a booming market. It was worth $1,217.1 billion in 2019 and will reach $3,206.3 billion in 2030. Therefore, it is a stable and growing industry. But most importantly, the pharma industry is crucial for the well-being of humankind. Where would we be now without anti-biotics, painkillers, vaccines, and other life-saving drugs? Not that long ago, HIV was a deadly disease, whereas people can get old with it nowadays. There are countless examples of how the pharmaceutical industry changed our lives in the past dozen years. And the most amazing fact is that the quality of the drugs is becoming only better and better.
Reputation Problems
The problem is that the pharmaceutical industry has gotten a bad reputation recently. When people hear Big Pharma, they directly think about some evil executives. Villains who sell medicine for overpriced value stash the money in their pockets. Then, hop in their private jets and go sip cocktails on a beach somewhere while the poor suffer. I won’t claim that some CEOs earn too much money. However, I will argue that medicine prices are not always overpriced, nor are they randomly set, only to allow the CEO to buy his third Lamborghini.

I won’t claim that some CEOs earn too much money. However, I will argue that medicine prices are not always overpriced, nor are they randomly set, only to allow the CEO to buy his third Lamborghini.
When people look at the prices of medicine, they rarely consider the vast amount of money and time that companies invest in new drug development. According to a study published in the journal JAMA, the cost to develop new pharmaceutical drugs is on average $1,3 billion. On top of that, it takes between 10 to 15 years to bring a new drug entity to the market.
The Drug Development Process
The process of developing new drugs has five stages. The first stage is called the drug discovery phase. In this stage, scientists test from 10 thousand to 15 thousand compounds or molecules for function through insights into disease processes and molecular testing. From those thousands of compounds, only about 250 show some promise and move to the next phase – Drug Development. In this phase, which lasts three to six years, the drugs researchers test, mainly for toxicity. They do it on cell lines, what’s called in-vitro, and on selected animal species, what’s called In-Vivo.
Only five candidates would do well in this phase and continue to the next stage- clinical trials. The trials last six to seven years. Statistically, only one drug candidate will prevail and move forward to the fourth step, which is a new drug application. If the drug is approved and goes to the market, it will be regularly monitored for safety by authorities. The monitoring is the fifth and last phase of the process and is called post-market drug safety monitoring.

The Patent Law
So, as you have seen, the road to a new drug is long and costly. From about 15,000 compounds, only one would reach the market. It would take 15 years, costing about 2.5 billion dollars.
Therefore, for a business to sustain itself and have an incentive to develop new innovative drugs, it must have a return for the invested billions, and then some, to thrive. Don’t forget about the production costs and all the employees that have to get their paychecks!
Some of you may still think: “yeah, you are right, but I still feel that the pharma companies rip us off.” They can sell the drug infinitely now, asking for a large amount of money. Thus, earning much more than what they have invested in the first place.
My reply to it would be a NO! They can’t because here comes to play small a thing which is called a Patent. A patent is a legal defense system. It gives its owner the legal right to exclude others from making or selling their invention for 20 years. But the situation is even less colorful. A company would usually apply for the patent already during the drug discovery phase. That means that by the time the company gets the drug approved for the market, it would have only ten years left to sell the drug as a monopoly. Some countries have law technicalities, which allow them to extend a drug patent for a few more years. I won’t get into it to not complicate things.
In summary, a pharma company gets ten years to be the sole manufacturer of a drug that took them another ten years to develop.

Getting the Recipe for the Drug
If you are new to patent law, you probably ask yourself, how do other companies know how to produce the same drug? Do they steal the recipe?
So – no! When a pharma company applies for the patent, it must describe precisely how its drug works and what makes it unique. Therefore, it becomes public knowledge, and others have the entire blueprint. Therefore, a patent is a double-sided sword.
The inventor protects his invention for a short period, but afterward, everybody can use it.
In short, a patent is an incentive to be the first to discover new things while benefiting the greater good by disclosing your secrets.
Two Types of Pharmaceutical Companies
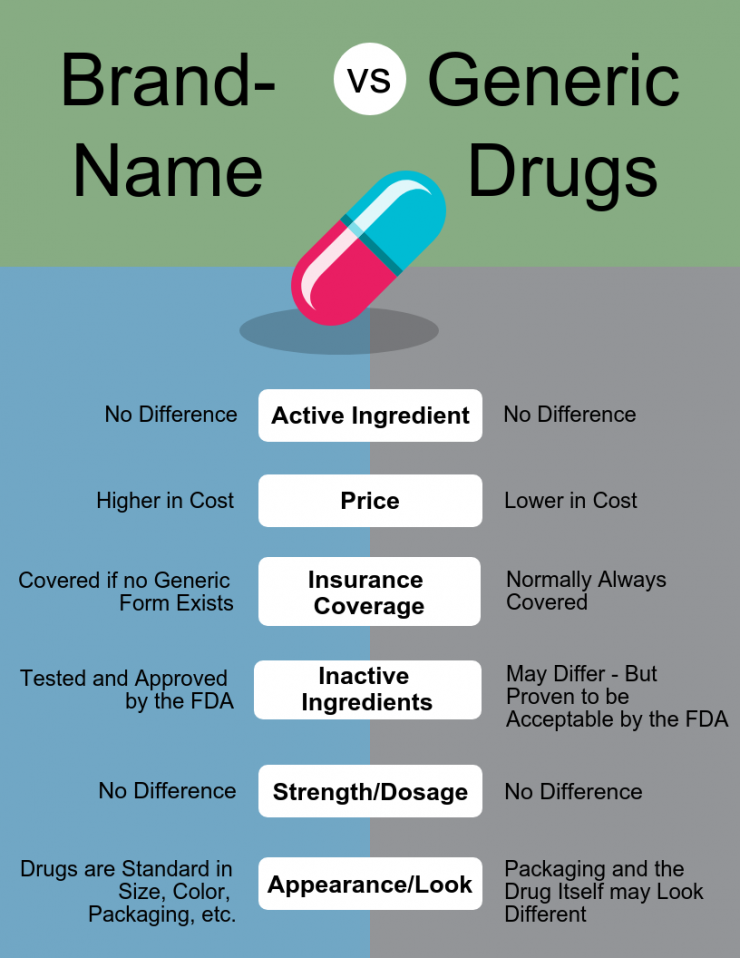
So, as mentioned, as soon as a patent expires, other companies may sell the same product. When we talk about pharma companies, we have to distinguish here between two types of those. The innovative pharmaceutical companies invest a lot of money and time in the research and development of new drugs but then rip the high returns for about ten years. On the other side, we have Generic Pharmaceutical Companies. Those wait until patents are expired and then manufacture the same drugs without investing much time and money in research. Generic pharma companies only have to show that their drug has the same active ingredients: quality, effectiveness, benefits, strength, and safety.
Sometimes the generic pharma companies don’t even wait for patents to expire but actively try to make a patent invalid with an army of attorneys.
It sounds wicked because the innovative companies invest tons of money in drug development, and then their rights are being snatched from them.
But it is legal and benefits the consumer, who gets to buy the drug for much cheaper, much earlier.
Drug Prices After the Loss of Exclusivity
For example, let’s look at a drug called Tamiflu, which Roche developed and released to the market in 1999. The price yearly revenue during exclusivity peaked at $750 million. As soon as several generic drugs entered the market in 2017, the revenue dropped to $200 million. We are talking about a drop of 73%! Therefore, if you get a subscription from your doctor for any medicine, make sure to ask the pharmacist if they have the same drug from a generic company. Doing so could save you much money.
At this point, you should understand the dynamics of the pharmaceuticals market and the reason for the initial high costs of medicine. What do you think about the medicine prices after reading this article?
Leave your thoughts in the comment section below.
Yan is the founder and managing director at Qualistery GmbH. Through his vision, GxP professionals worldwide can stay updated with current industry trends without breaking the bank.







